

When the Food and Drug Administration coaxed six states into scheduling kratom over three years, it drew a line in the sand for the industry. Advocates could get in line and try to adhere to a legal structure that went against science and customers’ demands or fight back. In the years since those bans, advocates have fought off a proposed federal ban and established regulated markets in nearly a dozen states.
The domino that kratom advocates haven’t quite been able to topple, though, is undoing the work that was encouraged by the FDA. Vermont may be on the verge of giving that effort a big push.
The American Kratom Association has been the driving force behind kratom regulations across the country, and it appears the organization is set for a breakthrough after an update from Vermont’s Department of Health. In an update sent out by the AKA earlier this year, the organization acknowledged that Vermont’s DOH “granted the petition” of the agency to remove kratom’s active alkaloids from the state’s Regulated Drug Rule, in essence overturning the ban enacted by the state legislature seven years ago.
Any action in the six states that banned kratom is considered a major win, especially considering that “the FDA has vigorously engaged in a disinformation campaign for more than a decade,” per the AKA’s update in April. The AKA received word from Vermont’s DOH on March 1 that “this email is to appraise you that the Department is granting your petition to remove mitragynine and 7-hydroxymitragynine from the Regulated Drug Rule.”
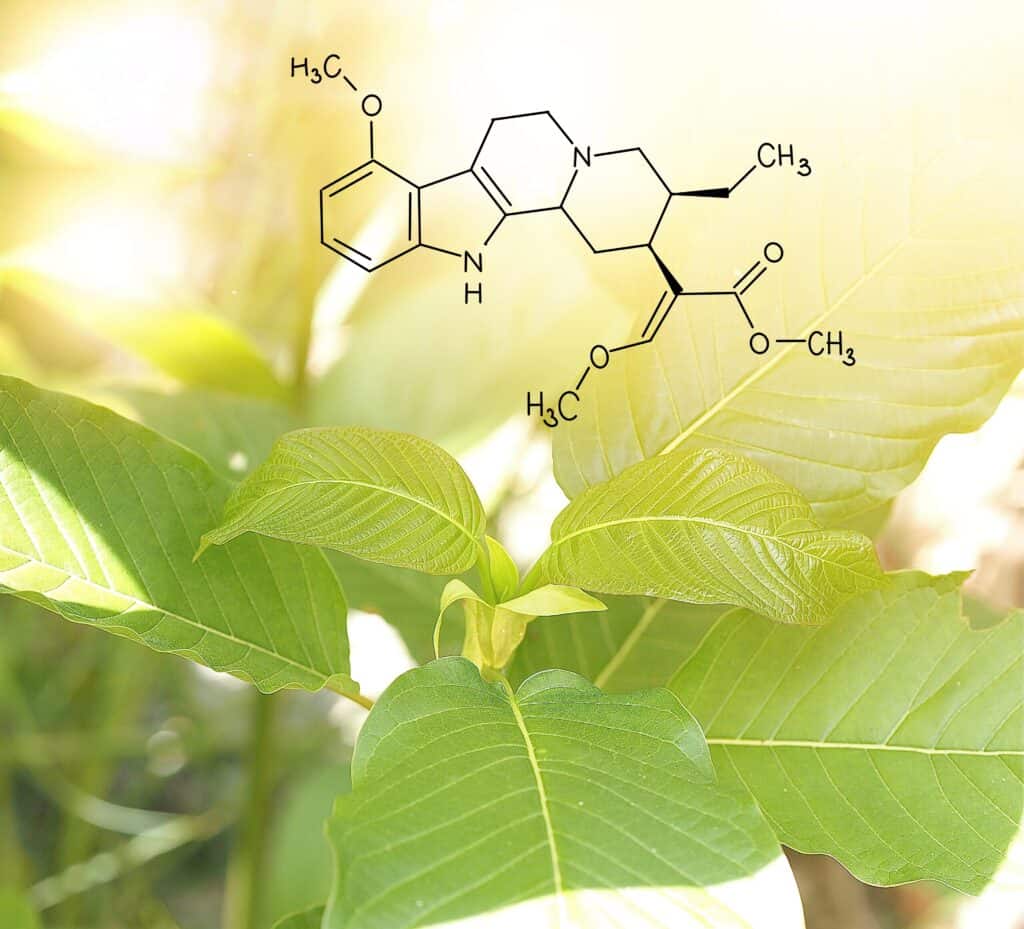
That was welcome news for the AKA, who contends the FDA’s actions were contrary to “tens of thousands of foods, dietary ingredients, botanical supplements and dietary supplements” similar to kratom. The science is clear, said the AKA, kratom is “not an opioid by plant genetics, by chemical structure, or by legal definition.”
In pretty much any way you can classify kratom, the AKA contends available data suggests the best course of action is establishing a safe, regulated market for the plant.
“The short answer is because the FDA is wrong on the science and wrong on the policy,” the AKA update said. “Other federal and international agencies have carefully evaluated the FDA’s claims and they find they lack sufficient evidence to support the FDA claims.”
Furthermore, the AKA contends that available studies, data and research on kratom call into question the FDA’s position on kratom’s risk and potential for addiction. When it comes to the science and facts on kratom, the AKA said all factors point toward the same conclusion.
“Kratom is safely used by consumers for a variety of purposes…typically as a replacement for a cup of coffee,” said the AKA’s update.

Vermont’s initial scheduling of kratom’s active alkaloids came in 2016, as part of a string of six states that took action against the plant in anticipation of pending federal action. The only problem is that the federal follow-up never arrived. Once the Drug Enforcement Agency announced its intention to schedule kratom, public backlash that included support from the scientific community quickly reversed that decision.
A second attempt by the FDA to encourage the DEA to schedule kratom also fell flat, this time with an admonishment from leadership within the Department of Health and Human Services.
That didn’t matter in Vermont, though, as the law passed by state legislatures didn’t include any connection to proposed federal action. When the federal ban fell through, Vermont kratom customers lost access to purchase a dietary ingredient that was still legal in 44 of the 50 states.
In the email from the AKA, the Vermont DOH said that rulemaking would commence shortly to complete the process. A reversal of Vermont’s ban on kratom could be the start of a significant shift for the states that took action on the FDA’s recommendation.
Rhode Island and Indiana represent the hope for states who looked to legislative solutions to past bans on kratom.
In Rhode Island, a comprehensive Kratom Consumer Protection Act was proposed by state legislators. At a hearing for that bill, lawmakers openly testified that the state was misled by the FDA when it banned kratom. That bill was unsuccessful this session but started a conversation that had remained dormant since the state scheduled kratom. Discussions around the proposed regulations even drew an acknowledgment from Rhode Island’s interim Director of the Rhode Island Department of Health to acknowledge that kratom does not meet the criteria for scheduling under state law.
Indiana lawmakers also took a run at overturning the kratom ban, with their version of a KCPA passing through the state’s House of Representatives by a vote of 54-30.
Advocates in Wisconsin, another state that banned kratom, are also on a similar path to Vermont with the Wisconsin Controlled Substances Board currently considering a report from local researchers. Per the AKA, the report to the CSB was accompanied by a conclusion from Dr. Chris Cunningham, an associate professor at Concordia University Wisconsin.
“Based on our review of the available literature, we conclude that regulation of M. speciosa in Wisconsin as a schedule-I substance is not justified at this time,” Cunningham’s report said. “We believe that controlling M. speciosa and its chemical constituents under schedule I harms public health.”









These statements and products presented on this website have not been evaluated by the Food and Drug Administration FDA. The products mentioned on this website are not intended to diagnose, prevent, treat or cure any diseases or health conditions. Therefore any information on this website is presented solely as the opinions of their respective authors who in which do not claim in any way shape or form to be medical professionals providing medical advice. SuperSpeciosa.com and its owners or employees cannot be held responsible for, and will not be liable for the inaccuracy or application of any information whatsoever herein provided. By purchasing our products you agree that you are aware and in compliance with your local county, state, or federal regulations. Must be 21 years or older to purchase Kratom. The US FDA has not approved kratom as a dietary supplement. We do not ship to the following states, cities and counties in the US where Kratom is banned: Alabama, Arkansas, Indiana, Rhode Island, Vermont, Wisconsin, Sarasota County, FL, Union County, MS, San Diego, CA, Jerseyville, IL, Oceanside, CA, and Ontario, OR. Furthermore, Kratom is also banned in the following countries where shipment cannot be executed: Australia, Burma, Denmark, Finland, Israel, Lithuania, Malaysia, Myanmar, Poland, Romania, South Korea, Sweden, Thailand, United Kingdom, Vietnam.
This product should be used only as directed on the label. It should not be used if you are pregnant or nursing. Consult with a physician before use if you have a serious medical condition or use prescription medications. A Doctor’s advice should be sought before using this and any supplemental dietary product. All trademarks and copyrights are property of their respective owners and are not affiliated with nor do they endorse this product. By using this site, you agree to follow the Privacy Policy and all Terms & Conditions printed on this site. Void where prohibited by law.
† These statements have not been evaluated by the FDA. This product is not intended to diagnose, treat, cure or prevent any disease.
Testimonials on this website are not intended as claims that our products can be used to diagnose, treat, cure, mitigate or prevent any disease. Read full disclaimer.


By submitting you agree to receive automated promotional messages. This agreement is not a condition of any purchase. See Terms and Privacy Policy. Frequency varies. Can opt out at any time.

There’s always something new happening. Enter your info below to get updates on huge deals, new products and special batch kratom releases.
By submitting you agree to receive automated promotional messages. This agreement is not a condition of any purchase. Message and data rates may apply. See Terms and Privacy Policy. Frequency varies. Can opt out at any time.