

With the number of states offering protection for kratom consumers now in double digits, Rhode Island was looking to be the first to flip a ban into legislation establishing a regulated kratom market with a bill that made its way through the state’s House of Representatives.
Unfortunately for consumers in Rhode Island, that effort will have to wait until next year due to the end of the legislative session.
The attempted flip of Rhode Island’s kratom policy fell flat when it failed to make it through committee by the deadline.
Matching bills were introduced in both the House of Representatives and Senate in Rhode Island, in hopes of overturning a ban on kratom that was enacted in 2017. Momentum briefly stalled when the Senate version was held for consideration, however, the recent House vote temporarily reignited hope that the state can find a solution for consumers who have incorporated the plant into their routine
That hope of overturning the ban on kratom relied on the fate of H5330, a bill introduced by a pair of Democratic legislators in hopes of establishing a regulated kratom market in a state where the active alkaloids of the plant are currently Schedule I substances. Rep. John Edwards and Rep. Brian Kennedy introduced the House version, to accompany a companion bill introduced by a trio of state senators.
The proposed legislation would have flipped Rhode Island from a state that schedules kratom to one with a comprehensive Kratom Consumer Protection Act. Contained within H5330 were regulations for manufacturers, restrictions on what can be included in kratom products and labeling instructions. Unlike other KCPAs, the Rhode Island version even went as far as to require processors to register annually and receive a third-party certificate of compliance with safe manufacturing standards.
That two-tiered approach of introducing matching bills in each chamber looked to be stalled out, when the House Corporations Committee decided to hold H5330 for further study the first week of April, then the Senate version of the bill was held by the committee a month later. Hopes surged to life earlier in June, though, when the House version found its way back onto the schedule.

At that point, the committee made a simple suggestion of raising the minimum age requirement to 21 years old, and that substitute was placed on the House calendar for a vote.
Due to the current restrictions on kratom in the state, the vote was closer than it has been on other KCPAs, but the measure still passed the Rhode Island House of Representatives by a vote of 44-23, including eight representatives that did not register a vote. Upon being sent to the Senate, the bill was steered toward the same committee that held the Senate version of the bill for further study.
With the attempt officially stalling out, one of the most robust back-and-forths on kratom policy in 2023 will most likely continue into next year.
Other unique aspects of the bill were requirements of processors to take immediate action in the wake of a “non-compliance report” and sets up a structure to report adverse events from kratom use.
During the hearing for the bill, both Edwards and Kennedy were adamant: This bill would help the state fix a piece of misguided legislation. According to the legislators, the only reason that kratom was scheduled in the first place came from undue influence by the Food and Drug Administration, with the states who took action left on their own when the federal government refused to schedule kratom, twice.
As more science has emerged, the representatives said early indications are that the greatest risk of serious reaction comes from illicit additives to kratom products. With kratom currently banned in the state, there is an increased risk of Rhode Island consumers being exposed to tainted kratom on the black market. Edwards and Kennedy, along with others testifying in favor, made it clear–access to pure, unadulterated kratom products was the best path forward.
At the center of the debate is the issue of kratom’s usage, as well as the safety profile of the plant. One of the key opponents to efforts to flip kratom’s legality is the RI Department of Health, who submitted written testimony against the bill. Some may question RIDOH’s stance, given that they were the organization that worked with the FDA to convince legislators to restrict kratom in the first place.
Among a variety of concerns listed were repeated claims by the FDA, as well as the claims that vendors online are illegally marketing kratom as a treatment option, despite no such uses being approved in the United States.
“Rhode Island cannot allow unscrupulous vendors to take advantage of consumers by selling products with unsubstantiated claims,” read the letter.
The only issue with that resistance is that the proposed legislation seems to address the same issue. Under H5330, kratom products would be defined as a food product and dietary ingredient, and require specific labeling and usage instructions that would be required by law to adhere to FDA guidelines.

In the letter from RIDOH, the agency only mentions kratom’s potential as a food ingredient once, saying “the current evidence regarding side effects and potential for adverse consequences associated with kratom use are sufficient to conclude it should not be available as either a food or supplement.” The letter does not include any direct references or evidence to back the department’s claims.
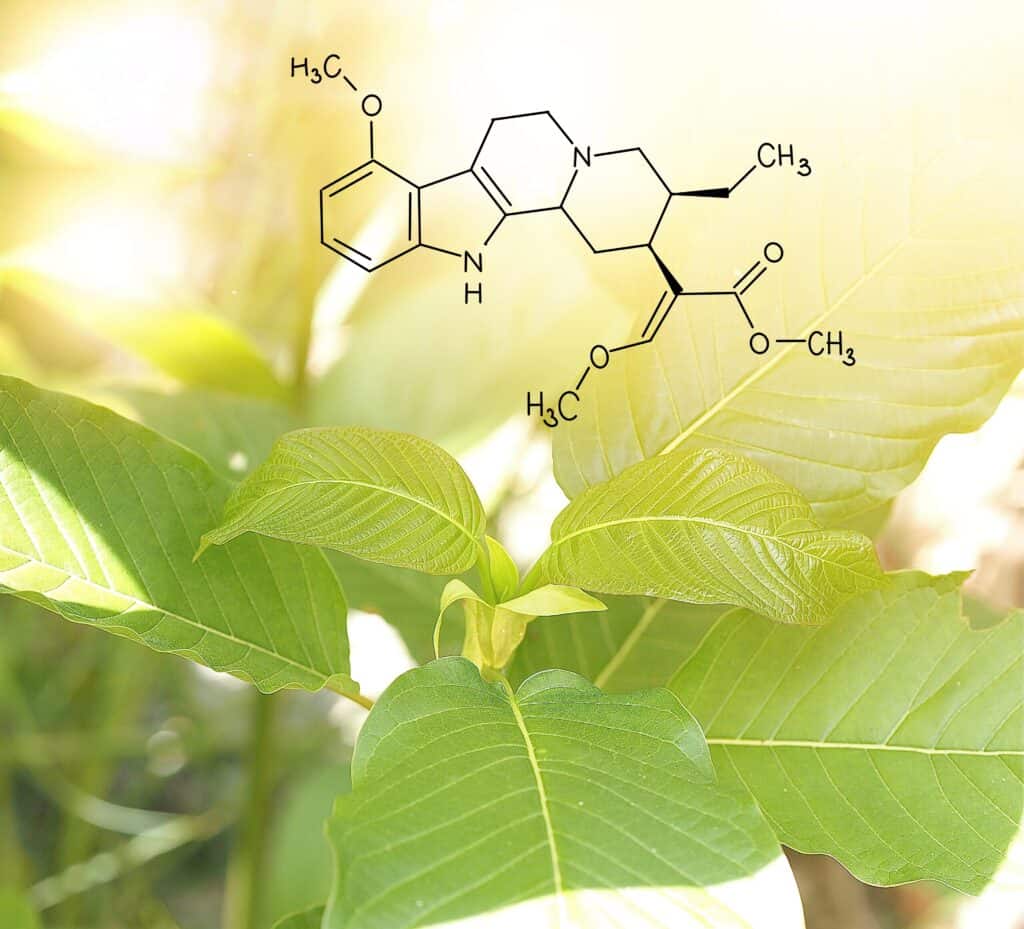
According to the National Institute on Drug Abuse, unadulterated kratom use alone does not present the same serious risk as classical opioids, with most serious side effects occurring as a result of polysubstance abuse and illicit substances that are included in adulterated kratom products. In fact, there are numerous ways that science is showing the differences between kratom and opioids.
Kratom is not an opioid, as it distinguishes itself from opioids in several key ways, particularly when it comes to side effects. Unlike typical opioids, kratom has been found, in initial animal studies, not to induce respiratory depression. Additionally, another notable difference is that kratom does not trigger the release of the Beta Arestin Protein, which is characteristic of conventional opioids. This directly contradicts points made in the RIDOH letter opposing a regulated kratom market.
It is one among the numerous methods through which scientific research has demonstrated that kratom is not as harmful as portrayed by these lawmakers. The American Kratom Association consistently promotes this fact, supported by evidence rooted in scientific findings, that pure kratom is safe for consumption.









These statements and products presented on this website have not been evaluated by the Food and Drug Administration FDA. The products mentioned on this website are not intended to diagnose, prevent, treat or cure any diseases or health conditions. Therefore any information on this website is presented solely as the opinions of their respective authors who in which do not claim in any way shape or form to be medical professionals providing medical advice. SuperSpeciosa.com and its owners or employees cannot be held responsible for, and will not be liable for the inaccuracy or application of any information whatsoever herein provided. By purchasing our products you agree that you are aware and in compliance with your local county, state, or federal regulations. Must be 21 years or older to purchase Kratom. The US FDA has not approved kratom as a dietary supplement. We do not ship to the following states, cities and counties in the US where Kratom is banned: Alabama, Arkansas, Indiana, Rhode Island, Vermont, Wisconsin, Sarasota County, FL, Union County, MS, San Diego, CA, Jerseyville, IL, Oceanside, CA, and Ontario, OR. Furthermore, Kratom is also banned in the following countries where shipment cannot be executed: Australia, Burma, Denmark, Finland, Israel, Lithuania, Malaysia, Myanmar, Poland, Romania, South Korea, Sweden, Thailand, United Kingdom, Vietnam.
This product should be used only as directed on the label. It should not be used if you are pregnant or nursing. Consult with a physician before use if you have a serious medical condition or use prescription medications. A Doctor’s advice should be sought before using this and any supplemental dietary product. All trademarks and copyrights are property of their respective owners and are not affiliated with nor do they endorse this product. By using this site, you agree to follow the Privacy Policy and all Terms & Conditions printed on this site. Void where prohibited by law.
† These statements have not been evaluated by the FDA. This product is not intended to diagnose, treat, cure or prevent any disease.
Testimonials on this website are not intended as claims that our products can be used to diagnose, treat, cure, mitigate or prevent any disease. Read full disclaimer.


By submitting you agree to receive automated promotional messages. This agreement is not a condition of any purchase. See Terms and Privacy Policy. Frequency varies. Can opt out at any time.

There’s always something new happening. Enter your info below to get updates on huge deals, new products and special batch kratom releases.
By submitting you agree to receive automated promotional messages. This agreement is not a condition of any purchase. Message and data rates may apply. See Terms and Privacy Policy. Frequency varies. Can opt out at any time.