

A roller coaster ride of a legislative session came to an end for kratom advocates in Virginia after the governor signed protections for kratom into law.
With the signature of Gov. Glenn Youngkin, kratom is now included in Virginia’s Consumer Protection Act, including guidelines for age requirements and labeling. At the same time, the amendment that passed was substantially different from what was proposed, leaving work to be done to match other states that have passed kratom regulations.
The bill, as originally introduced by Delegate Hyland “Buddy” Fowler, proposed to add an amendment to a section of the Virginia Code that is commonly referred to as the Virginia Consumer Protection Act. In Fowler’s version, the amendment would have penalized anyone selling adulterated or synthetic kratom and set forth strict manufacturing and product standards for consumers over the age of 18.
It appeared the bill had momentum when it was passed through the Senate by a vote of 39-0, before being handed off to the Committee on General Laws upon its arrival in the state’s House of Representatives.
However, it was met with resistance by multiple delegates who referenced the federal government’s position on kratom as a cause for taking a pause. Delegate Danica Roem spoke in opposition to the bill and cited a report from the Food and Drug Administration about the risks associated with kratom.
Roem’s argument had an impact on the committee, which voted against the measure as it was written by a vote of 14-7. At the time, a delegate on the committee referenced the federal regulatory agency in comments about why the bill stalled as written.
“I feel like the best action at this time is either as a state we study this in a workgroup and figure out what those regulations would look like and what we’re concerned about or we do nothing and let the FDA give us some guidance,” said Delegate Dawn Adams.
From there, the bill was rewritten to accomplish two things: Raising the age of purchase to 21, and requiring a label with specific guidance.
“This product may be harmful to your health, has not been evaluated by the FDA, and is not intended to diagnose, treat, cure, or prevent any disease.”
With a rewritten bill in place of the original, both houses of the Virginia legislature unanimously passed the bill, and it was signed by Youngkin the day before the deadline for his signature. Even with the changes, advocates in the state and at the national level are happy that a dialogue has begun.
Virginia resident Sage Giles told local news channel WRIC that any sort of law establishing a structure for kratom sales is welcome news to kratom users like herself.
“It’s not perfect but I love that this is a starting point and will make people think,” Giles said. “I think with the proper labeling, it will be a lot easier.”
Representatives from the American Kratom Association initially told WRIC they were happy that Fowler’s version would address the issue of adulterated kratom. With the changes to the bill, the AKA acknowledged “it helps” but said it leaves work to be done.
“We hope to come back and strengthen this law later.”
The frustrating part for advocates is the role the FDA continues to play in clouding these discussions. Those who have followed the legal efforts to regulate kratom in America are familiar with the adversarial nature of the FDA toward the kratom industry. Despite a failed attempt to encourage the Drug Enforcement Administration to schedule kratom, the FDA has continued to issue import alerts and consumer notices warning against kratom, despite questionable science behind those claims.
It’s gotten to the point that a bipartisan group of legislatures introduced federal legislation to release available research on kratom and stop the FDA from taking further action without justifiable cause.
Nine other states have adopted versions of what advocates call the Kratom Consumer Protection Act (KCPA), which is similar to the original amendment proposed by Fowler. In those states, regulations have required QR codes with batch/product information. This means strict manufacturing and label standards that include identifying all ingredients. To ensure complete safety, some KCPA’s mandate independent lab testing to ensure access to safe, unadulterated kratom.
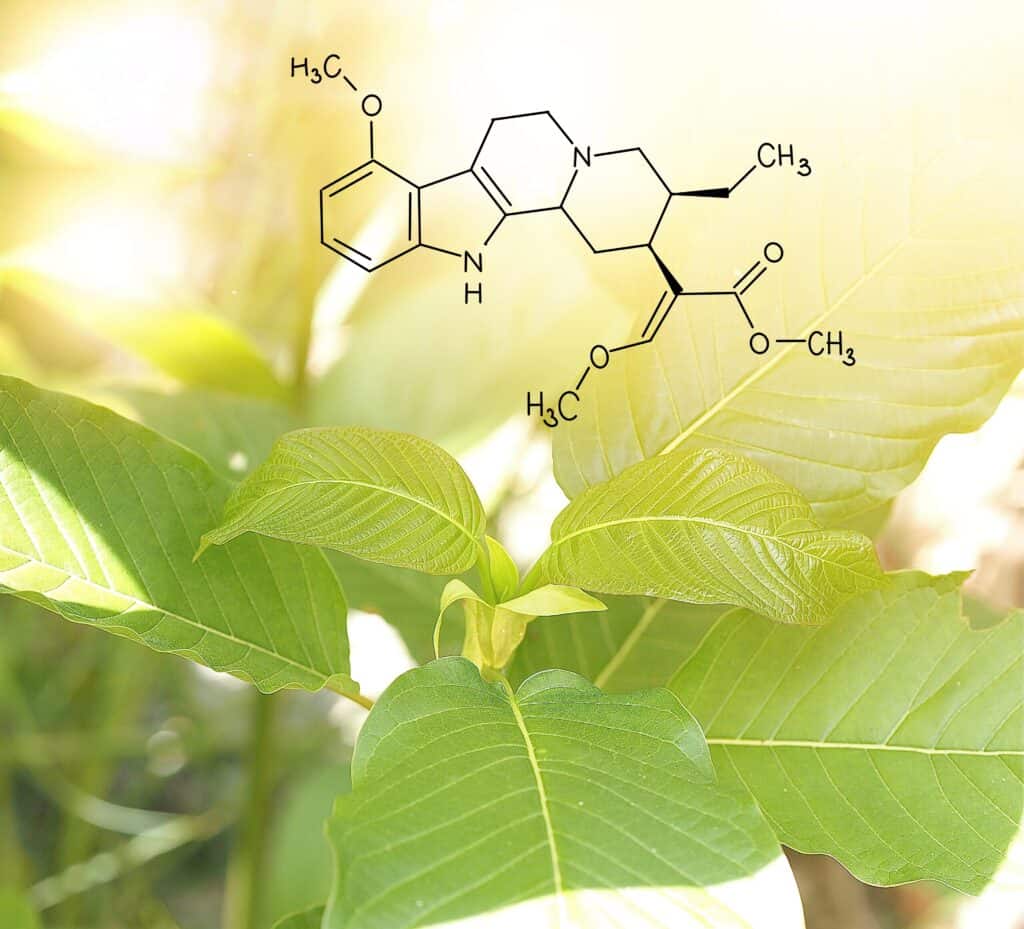
Fowler’s version of the amendment called for specifics such as banning “any kratom extract that contains levels of residual solvents than is allowed” by medical standards, and even sets a ceiling for the concentration of kratom in a product. It also set a more specific definition of kratom and subsequent terms, but that section was also simplified down to kratom being defined as “any part of the leaf of the plant Mitragyna speciosa or any extract thereof.”
In other states, advocates and lawmakers who sponsored similar legislation have been willing to negotiate with state health officials on such topics as raising the minimum age of purchase from 18 to 21. In this instance, a committee instead decided to rewrite the proposal and opted for an approach that simply sets an age requirement and a warning label for a product that was already legal to buy in the state.









These statements and products presented on this website have not been evaluated by the Food and Drug Administration FDA. The products mentioned on this website are not intended to diagnose, prevent, treat or cure any diseases or health conditions. Therefore any information on this website is presented solely as the opinions of their respective authors who in which do not claim in any way shape or form to be medical professionals providing medical advice. SuperSpeciosa.com and its owners or employees cannot be held responsible for, and will not be liable for the inaccuracy or application of any information whatsoever herein provided. By purchasing our products you agree that you are aware and in compliance with your local county, state, or federal regulations. Must be 21 years or older to purchase Kratom. The US FDA has not approved kratom as a dietary supplement. We do not ship to the following states, cities and counties in the US where Kratom is banned: Alabama, Arkansas, Indiana, Rhode Island, Vermont, Wisconsin, Sarasota County, FL, Union County, MS, San Diego, CA, Jerseyville, IL, Oceanside, CA, and Ontario, OR. Furthermore, Kratom is also banned in the following countries where shipment cannot be executed: Australia, Burma, Denmark, Finland, Israel, Lithuania, Malaysia, Myanmar, Poland, Romania, South Korea, Sweden, Thailand, United Kingdom, Vietnam.
This product should be used only as directed on the label. It should not be used if you are pregnant or nursing. Consult with a physician before use if you have a serious medical condition or use prescription medications. A Doctor’s advice should be sought before using this and any supplemental dietary product. All trademarks and copyrights are property of their respective owners and are not affiliated with nor do they endorse this product. By using this site, you agree to follow the Privacy Policy and all Terms & Conditions printed on this site. Void where prohibited by law.
† These statements have not been evaluated by the FDA. This product is not intended to diagnose, treat, cure or prevent any disease.
Testimonials on this website are not intended as claims that our products can be used to diagnose, treat, cure, mitigate or prevent any disease. Read full disclaimer.


By submitting you agree to receive automated promotional messages. This agreement is not a condition of any purchase. See Terms and Privacy Policy. Frequency varies. Can opt out at any time.

There’s always something new happening. Enter your info below to get updates on huge deals, new products and special batch kratom releases.
By submitting you agree to receive automated promotional messages. This agreement is not a condition of any purchase. Message and data rates may apply. See Terms and Privacy Policy. Frequency varies. Can opt out at any time.