
Amidst a back-and-forth year of legislative efforts centered around kratom, advocates are hoping that a bill in the Rhode Island legislature can deliver a gut punch to opponents seeking to ban kratom, and at the same time set the record straight on the science surrounding the substance.

A pair of state Representatives introduced H5330 earlier this year, a bill aimed to overturn a 2017 decision by Rhode Island that scheduled kratom as a Schedule I. According to the sponsors of the bill, state officials were previously swayed by misleading information from the Food and Drug Administration, and the two representatives are aiming to right that “wrong” and give kratom consumers in Rhode Island access to a safe, regulated market.
Rep. Brian Patrick Kennedy is one of the sponsors, and also a member of the House Committee on Corporations, who heard testimony on the proposed bill earlier this month. For Kennedy, this bill is about doing right by his constituents.
“What I would point out is that from 2012-2017 the FDA tried to get states to ban kratom,” Kennedy said. “Very few states decided to do that, Rhode Island, unfortunately, was one of those states.”
“Kratom ban has been in place in six states up to this point in time, so that meant 44 states did not fall for the ploy of the FDA,” he said. “Vermont recently announced that they were going to deschedule it… that leaves Rhode Island as the only state (in New England) where you can’t buy kratom.”
Due to that availability in nearby states, Kennedy said it was time to listen to available science and provide a safe, fiscally responsible option for Rhode Island kratom consumers. Especially considering that nearby states had not yet enacted what is known as a Kratom Consumer Protection Act (KCPA), Kennedy said establishing a kratom market with labeling and manufacturing standards would be a net positive for the state in a variety of ways.
“What we’re looking to do is create a safe, unadulterated product of kratom that will be offered at retail locations around the state of Rhode Island,” Kennedy said. “This will be a revenue positive for the state of Rhode Island, in the fact that it will generate sales tax.”
The bill’s other sponsor, state Rep. John G. Edwards, echoed the same sentiments about the influence the FDA had on the process of scheduling kratom in the first place. Edwards even went as far as to reach out to the Department of Behavioral Healthcare, Development Disabilities & Hospitals (BHDDH) to clarify the most recent scientific information.
“They’re relying on a very very old FDA statute, they are not looking at the current information on kratom,” Edwards said. “And I reminded them as such, and they said they were still sending their letter through.”
That letter was presented by the committee as testimony in opposition to the bill, although no representative from BHDDH spoke in opposition at the hearing. The Rhode Island Department of Health (RIDOH) also submitted testimony in opposition and did not testify at the hearing.
Despite their opposing viewpoints, and RIDOH’s letter echoing similar sentiments to the FDA, Kennedy, and Edwards said they had productive meetings with members of the governor’s staff and the interim director of RIDOH. Among the topics discussed was raising the legal purchase age from 18, as proposed in the bill, to 21, as suggested by state officials.
That conversation also touched on practical matters of enforcement and penalties for violating the terms of the legislation.
“The Department of Health has expressed an interest in not having to regulate this because there currently isn’t a regulation process that they have available to them, so we are looking at the appropriate agency to put this in,” Kennedy said.
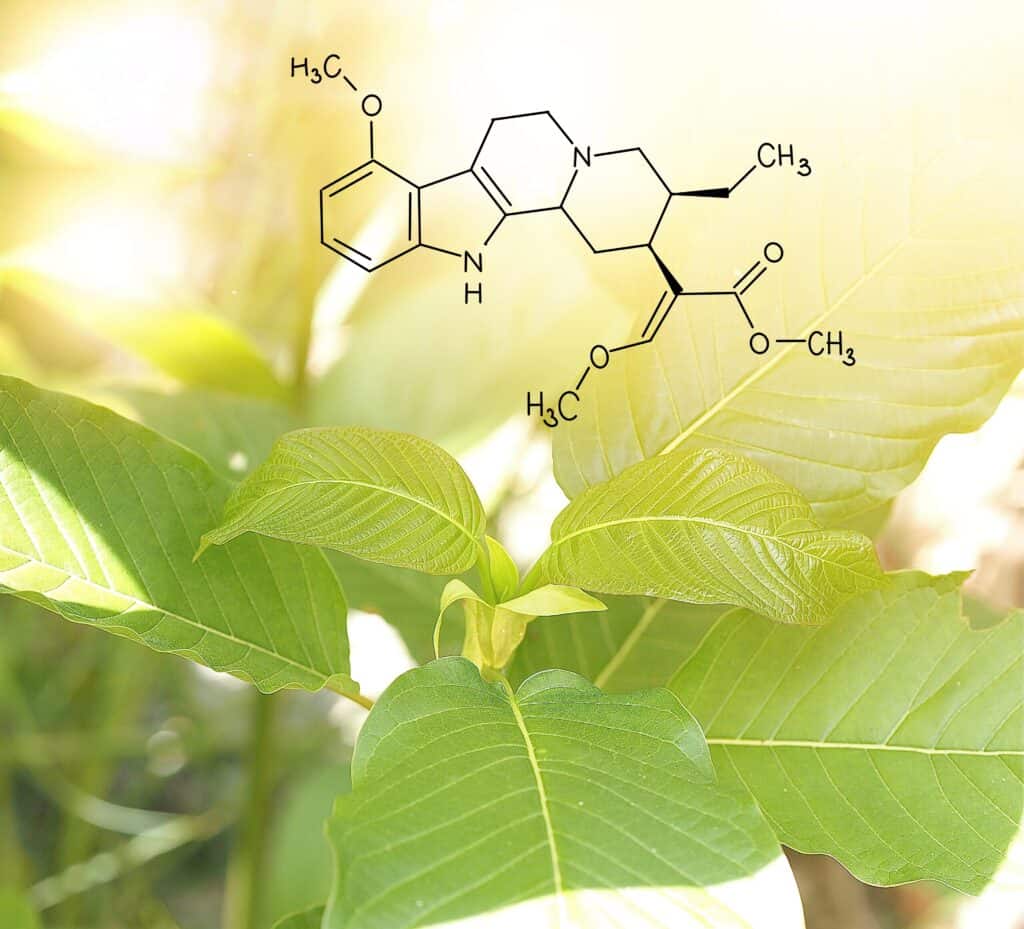
Mac Haddow, a senior policy fellow with the American Kratom Association, provided the data and scientific backing for the points made by Kennedy and Edwards and went into detail about what sets kratom apart from the classic opioids it is often incorrectly associated with. Not only did the FDA “get it wrong” on its recommendations to states, but Haddow also said there are developing studies and peer-reviewed articles that suggest the opposite of what is being claimed by the federal regulatory agency.
Haddow went on to express sympathy for families who have lost loved ones to suspected kratom overdoses but clarified that the science shows that adulterated products present the primary risk to consumers. He said the chemical profile of pure kratom suggests a lower risk of toxicity.
Questions from the committee ranged from cost to basic information about kratom and its use. The most focused set of questions came on kratom’s addiction potential, with Haddow stepping in to address the issue. While he acknowledged that kratom carries a potential for physical dependency, similar to other everyday substances, the prevailing science shows that the risk of physical addiction was relatively low.
As with most of the concerns around kratom, Haddow said those issues become especially amplified when there is a lack of a regulated market.
“(Vendors add) any number of products that will give what the natural kratom plant does not provide, which is a euphoric high,” Haddow said. “We don’t want that to be in the marketplace, we want people to have proper labeling and proper manufacturing standards.”









These statements and products presented on this website have not been evaluated by the Food and Drug Administration FDA. The products mentioned on this website are not intended to diagnose, prevent, treat or cure any diseases or health conditions. Therefore any information on this website is presented solely as the opinions of their respective authors who in which do not claim in any way shape or form to be medical professionals providing medical advice. SuperSpeciosa.com and its owners or employees cannot be held responsible for, and will not be liable for the inaccuracy or application of any information whatsoever herein provided. By purchasing our products you agree that you are aware and in compliance with your local county, state, or federal regulations. Must be 21 years or older to purchase Kratom. The US FDA has not approved kratom as a dietary supplement. We do not ship to the following states, cities and counties in the US where Kratom is banned: Alabama, Arkansas, Indiana, Rhode Island, Vermont, Wisconsin, Sarasota County, FL, Union County, MS, San Diego, CA, Jerseyville, IL, Oceanside, CA, and Ontario, OR. Furthermore, Kratom is also banned in the following countries where shipment cannot be executed: Australia, Burma, Denmark, Finland, Israel, Lithuania, Malaysia, Myanmar, Poland, Romania, South Korea, Sweden, Thailand, United Kingdom, Vietnam.
This product should be used only as directed on the label. It should not be used if you are pregnant or nursing. Consult with a physician before use if you have a serious medical condition or use prescription medications. A Doctor’s advice should be sought before using this and any supplemental dietary product. All trademarks and copyrights are property of their respective owners and are not affiliated with nor do they endorse this product. By using this site, you agree to follow the Privacy Policy and all Terms & Conditions printed on this site. Void where prohibited by law.
† These statements have not been evaluated by the FDA. This product is not intended to diagnose, treat, cure or prevent any disease.
Testimonials on this website are not intended as claims that our products can be used to diagnose, treat, cure, mitigate or prevent any disease. Read full disclaimer.


By submitting you agree to receive automated promotional messages. This agreement is not a condition of any purchase. See Terms and Privacy Policy. Frequency varies. Can opt out at any time.

There’s always something new happening. Enter your info below to get updates on huge deals, new products and special batch kratom releases.
By submitting you agree to receive automated promotional messages. This agreement is not a condition of any purchase. Message and data rates may apply. See Terms and Privacy Policy. Frequency varies. Can opt out at any time.